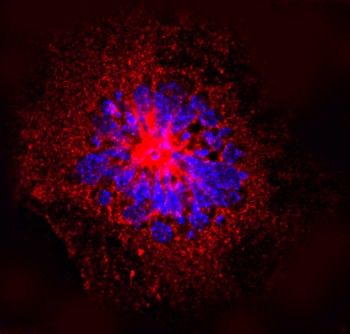
Endothelial cell. In red: immunolabelling of vimentin. In blue : nuclear DNA stained with DAPI. Credits: Scuola Superiore Sant’Anna, Pisa, Italy
Jessica Pietsch of the Spheroids project introduces the team’s experiment: the Spheroids project is a biological experiment with human cells found on the inner layer of blood vessels: the endothelial cells. Endothelial cells are in direct contact with the blood and regulate many things such as blood pressure for example.
Since astronauts show several alterations and dysfunctions during a space flight connected to blood pressure we put endothelial cells in simulated microgravity (s-µg) on Earth generated by a Random Positioning Machine that rotates samples continuously to create an average gravity sensation that is close to zero. Normally endothelial cells grow as single layers on the bottom surface of cell-culture flasks. In simulated microgravity we noticed the endothelial cells formed three-dimensional cell aggregates. These cell aggregates were tube-like and resembled a small, rudimentary, blood vessel. Combining the endothelial cells with other cultures we were even able to ‘rebuild’ several layers of a blood vessel.
In the Spheroids experiment we want to understand why and how the endothelial cells form these structures and adapt to microgravity. To achieve this we proposed the ESA Spheroids project to find out whether the findings in simulated microgravity are consistent with changes we expect to see in in real microgravity – in space on the International Space Station.
Experiment setup
The experiment will be conducted in experiment containers that operate automatically to cultivate the endothelial cells. Each Experiment Container includes compartments for cells to grow, the Cultivation Chamber, as well as compartments for with the cell nutrients (cell culture medium in medium chamber, MC) and fixatives to preserve each container after growth for later analysis back on Earth. Each Experiment Container contains two Cultivation Chambers and two cell-culture Medium Chambers. Before fixation, the culture medium is extracted from the Cultivation Chambers into their own Supernatant Chambers. The fixative is stored and injected from the Fixative Chamber.
There are two separate experiments (E1 and E2) in one Experiment Container so one cell-culture-medium can be supplemented with the protein Vascular Endothelial Growth Factor (VEGF). This protein has a protective influence on endothelial cells. E1 contains plain culture medium without VEGF while E2’s culture medium is supplemented with VEGF.
The table below summarizes the experiments performed in the Experiment Container during Principia. The duration of the experiments varies between a week to two weeks. During the 14-day experiment a change of growth medium occurs after 7 days.
To ‘fix’ or preserve the experiment for analysis we use paraformaldehyde (PFA) and RNAlater. The paraformaldehyde preserves any three-dimensional aggregates that might form for structural analysis on Earth. RNAlater preserves the cell RNA for gene analyses.
The experiment containers will go inside ESA’s Kubik incubator to keep them at the right temperature.
| Hardware | Experiment Side | Duration days | VEGF | Fixative Type |
| 1 | E1 | 7 | n | PFA |
| E2 | 7 | y | PFA | |
| 2 | E1 | 7 | n | RNAlater |
| E2 | 7 | y | RNAlater | |
| 3 | E1 | 14 | n | PFA |
| E2 | 14 | y | PFA | |
| 4 | E1 | 14 | n | RNAlater |
| E2 | 14 | y | RNAlater |



Discussion: no comments